Research Concept
In the transregional collaborative research center TRR 261 ‘Cellular Mechanisms of Antibiotic Action and Production’ (acronym ‘ANTIBIOTIC CellMAP’), we study antibiotic action and antibiotic production side-by-side within living bacteria, yet with a molecular focus. It is our vision to explore the cellular mechanisms of antibiotic action and production in space and time – thereby contributing to an ‘ANTIBIOTIC CellMAP’ – to simultaneously lay a foundation for more rational approaches in antibiotic drug discovery in the future and improve our understanding of fundamental principles of the cellular organization of prokaryotic life.
Over decades we have experienced a dwindling pipeline of new structural scaffolds in antibiotic drug discovery. This is not to the least due to substantial knowledge gaps in fundamental antibiotic research and lack of insight into bacterial cell biology. It became obvious that, even for long-established antibiotics, we often do not understand the cellular consequences of inhibiting a given target, although the direct ligand-target interactions are thoroughly studied. However, it is often the pleiotropic downstream effects following primary target interaction which cause efficient bacterial killing, such as the subsequent deregulation or disintegration of large biosynthetic machineries. Studying antibiotic action on the cellular level will allow us to differentiate potent from less potent antibacterial mechanisms, to elucidate the reasons for synergistic activities between antibiotics, and to explain specificities related to certain species or cell types. Likewise, with regard to antibiotic production, many antibiotic gene clusters and related enzyme functions are known. However, we know little about the localization and physical interactions within biosynthetic production machineries as well as about the physiology of the producer cell in the course of the production process. Studying the functional interplay between the various components of the biosynthetic machinery, its integration into cellular regulatory networks and cross-talk with the primary metabolism, as well as producer stress and adaptations, we aim towards a more rational and efficient production process.
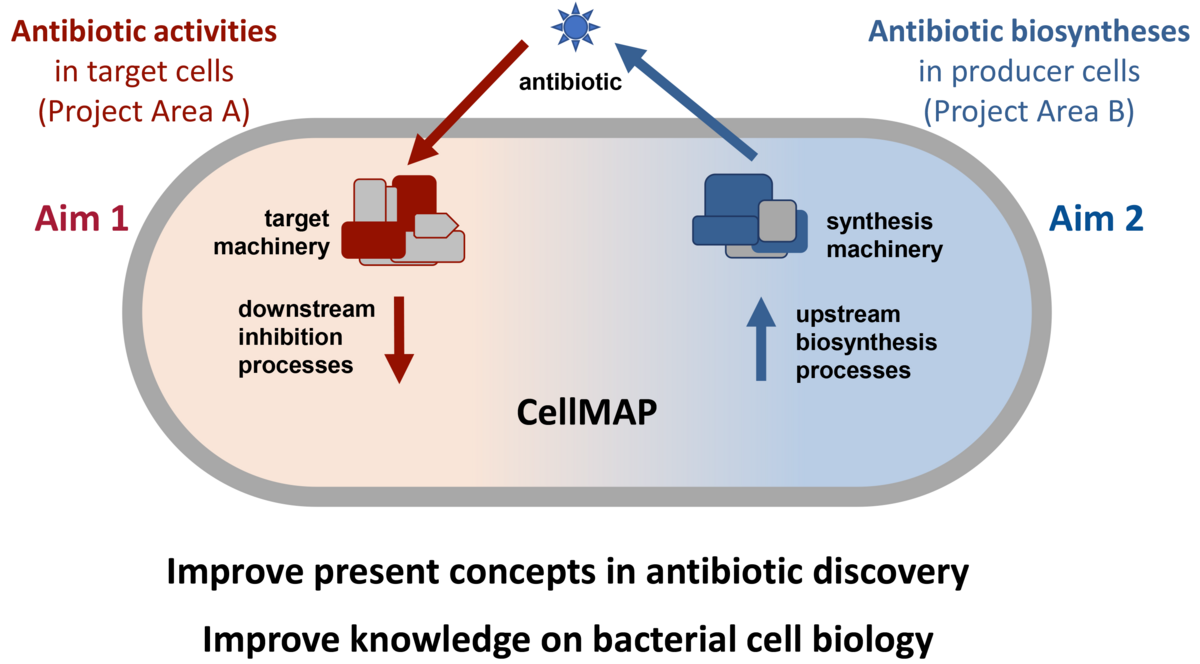
The overarching goals of this initiative are to provide insights into fundamental cellular processes associated with production and activities of antibiotics and to simultaneously enhance our knowledge of principles governing bacterial cell biology. Traditionally, it has been a highly successful research strategy in cell biology to use inhibitors for analyzing functional and organizational processes in living cells. As e.g., much of our understanding of peptidoglycan synthesis and ribosome function stems from previous investigations using antibiotic inhibitors, our studies will also increase our general knowledge about the cellular processes perturbed by the antibiotics studied here. In this initiative, researchers from microbiology, pharmacy, medicine, biochemistry, and chemistry will combine their multidisciplinary and complementary expertise to augment our general knowledge base on antibiotics, to improve present concepts and to lay a foundation for viable translational research and development activities in the future.

